Plastic Surgery Vascular Doppler Machine Supplier in India
PLASTIC SURGERY
HAND HELD VASCULAR DOPPLER
Minidop ES100VX
The ES-100VX Mini Doppler is a cost effective hand held pocket Doppler with integrated speaker for optimal sound sensitivity. Ideal for pedal pulses, systolic pressures and fetal heart sounds.
Features:
- Multi frequency 5,8,10 MHz Bidirectional Doppler
- 300 mW speaker output
- Power from DC9V, alkaline battery
- Conforms to IEC 60601-1
- Software for patient Report

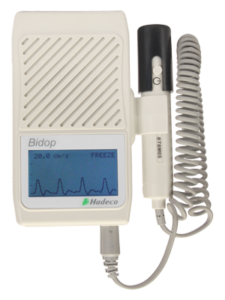
Bidop ES100V3
The Bidop ES100V3 is a visual bidirectional vascular pocket Doppler. View a real-time waveform on the LCD and store up to 30 waveforms for later reference or to download to Smart-V-Link vascular software. Options include a PPG probe for toe pressures and Smart-V-Link software.
Features:
- Bi-directional Doppler with backlight waveform LCD
- 30 waveform memory and USB interface
- Real-time waveform LCD -displays the waveform during Testing
- 30 waveform memory storage – save waveforms to view later or download to Smart-V-Link software
- Software for patient Report
- Automatic gain and baseline control – no manual adjustments required
- Vascular Link Software Smart-V-Link for Windows (option)
- Flat vascular probe available – eliminates the need to hold the probe during testing
- PPG available to measure toe pressure & study (option)
VRP PROBE 10MHz
VRP 10MHz Surgical probe is compatible with Hadeco Bidirectional Dopplers Minidop ES100VX, Bidop ES100V3, Smartdop45.

STERILISATION PROCEDURE
METHOD 1 (ETO)
CLEANING
Probes must be cleaned to remove contaminating substances prior to sterilization. After each use, perform initial cleaning within the sterile field by wiping the probe free of blood and body fluids. Then, pass the probe out of the field. Disconnect the probe (and any adaptor) from the Doppler unit, clean the probe tip and housing thoroughly with a mild soapy solution, a soft cloth with an enzymatic cleaning solution, or an alcohol-based wipe. If using an enzymatic cleaning solution, wipe the probe again using mild soapy water or an alcohol-based wipe. Do not soak. Complete preparation for sterilization in accordance to hospital procedure using an approved probe cleaning agent.
STERILIZATION 80%
It is recommended that the intraoperative and laparoscopic probes be gas sterilized before each use with ethylene oxide (ETO) gas. The suggested mixture ratio is ETO gas – 20%, CO2 gas – 80%. Exposure time should not be less than 1 hour and 45 minutes at a pressure of 0.75 to 0.8Kg/cm2 and a temperature of 50° to 60° C. To achieve a Sterility Assurance level (SAL) of 10-6 (the recommended minimum for direct contact with blood), an exposure time of 3 hours and 30 minutes is necessary. A biological indicator strip should be placed in the load to verify the effectiveness of the cycle selected.
WARNING
Do not sterilize the Doppler probes listed above by steam autoclaving. Steam sterilization is likely to damage the plastic probe tip and piezo-electric crystals. Utilize Koven/Hadeco probe number ACP-08 for steam autoclaving. Do not use an automatic washer/disinfector. Temperatures will cause probe electronics to melt.
AERATION
Follow hospital requirements with regard to aeration time to allow residual ETO gas to dissipate. The minimum recommended aeration time is 8 hours at 60° C, 40% to 60% humidity with an air change (ventilation) frequency of 25 times per hour. Note: aeration time should meet institution and FDA guidelines.
AERATION
During the examination, fluid coupling must be maintained for proper signal reception. If needed, irrigate the incision or laparoscopic portal with sterile saline to insure adequate coupling. Use of sterile gel is acceptable in all areas except the brain.
CONTRAINDICATIONS: Hospital policy takes precedence over this protocol.
Failure to use approved cleaning & sterilization procedures may void the warranty
METHOD 2 (STERRAD)
CLEANING
Probes must be cleaned to remove contaminating substances prior to sterilization. After each use, perform initial cleaning within the sterile field by wiping the probe free of blood and body fluids. Then, pass the probe out of the field. Disconnect the probe (and any adaptor) from the Doppler unit, clean the probe tip and housing thoroughly with a mild soapy solution, a soft cloth with an enzymatic cleaning solution, or an alcohol-based wipe. If using an enzymatic cleaning solution, wipe the probe again using mild soapy water or an alcohol-based wipe. Do not soak. Complete preparation for sterilization in accordance to hospital procedure using an approved probe cleaning agent.
STERRAD STERILIZATION SYSTEM GUIDELINES
The probes listed above may be sterilized with the Sterrad 100S, Sterrad 200, Sterrad NX and/or Sterrad 100NX (where indicated) sterilization systems (see charts above). The general processing instructions in the “Sterrad 100S, Sterrad 200, Sterrad NX and/or Sterrad 100NX Sterilization System Operator’s Manual(s)” should be followed. Probes must be cleaned to remove contaminating substances prior to sterilization. After each use, perform initial cleaning within the sterile field by wiping the probe free of blood and body fluids.
STERILIZATION
Per recommended Sterrad 100S, Sterrad 200, Sterrad NX and/or Sterrad 100NX procedures using a Sterrad brand or Sterrad compatible trays.
WARNING
Sterilize according to instructions for the Sterrad 100S, Sterrad 200, Sterrad NX, and/or Sterrad 100NX Model(s). Do not sterilize the Doppler probes listed above by steam autoclaving. Steam sterilization is likely to damage the plastic probe tip and piezo-electric crystals. Utilize Koven/Hadeco probe number ACP-8 for steam autoclaving. Do not soak or submerge the probe in liquid. Do not use Cidex. Do not use an automatic washer/disinfector. Temperatures will cause probe electronics to melt.
Failure to use approved cleaning & sterilization procedures may void the warranty.
 kodymedical@gmail.com
kodymedical@gmail.com +91-9380223396
+91-9380223396 My Account
My Account
